WATER DISTILLATION
A favored method of "Water Treatment" among many preppers, survivalists, homesteaders, and government entities is the process of Water-Distillation. Water Distillation kills organic material, sanitizes the container, and removes many chemical hazards. The process is a preffered way of treating almost any water source because distillation is the method of separating mixtures. Distillation is not a chemical reaction, but a unit operation or physical separation process. In short your separating the harmful elements from the H2O or in this case water vapor, which is created when you boil the source.
How it Works!
Distillation works by heating a liquid in a bowl. Chemicals turn to vapor at different temperatures, sometimes within a vacuum chamber. The vapor rises and collects on the sides of the upper chamber where it condenses back into a liquid and flows out through a spout. Thus the elements separate according to temperature, some turn to vapor at lower temperatures than others. Water Distillation uses the same process, but because Hydrogen and Oxygen separate at waters-boiling-point and many other chemicals do not, it becomes fairly easy to distill water from chemically saturated sources. In fact the water distillation process used on seawater will separate the hydrogen and oxygen from the salt, making seawater drinkable.
Distillation works by heating a liquid in a bowl. Chemicals turn to vapor at different temperatures, sometimes within a vacuum chamber. The vapor rises and collects on the sides of the upper chamber where it condenses back into a liquid and flows out through a spout. Thus the elements separate according to temperature, some turn to vapor at lower temperatures than others. Water Distillation uses the same process, but because Hydrogen and Oxygen separate at waters-boiling-point and many other chemicals do not, it becomes fairly easy to distill water from chemically saturated sources. In fact the water distillation process used on seawater will separate the hydrogen and oxygen from the salt, making seawater drinkable.
In History!
Distillation can be traced back to the 1st Century in Alexandria where Greek Alchemists employed the process in their experiments. Of course it is possible that one day we will find that distillation goes back even futher. A handful of archeological digs in southwest Asia indicate distillation may have been a process used to remove salt from water, (called desalination) about 5000 B.C.
Distillation can be traced back to the 1st Century in Alexandria where Greek Alchemists employed the process in their experiments. Of course it is possible that one day we will find that distillation goes back even futher. A handful of archeological digs in southwest Asia indicate distillation may have been a process used to remove salt from water, (called desalination) about 5000 B.C.
Illustration of Distillation Apparatus
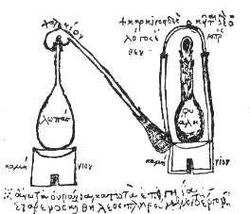
Distillation apparatus of Zosimus, from Marcelin Berthelot, Collection des anciens alchimistes grecs (3 vol., Paris, 1887–1888).
http://en.wikipedia.org/wiki/File:Zosimosapparat.jpg
Distillation played an important in the development of early sciences, just as it plays vital role in the manufacturing of thousands of products today. In the early days of alchemy, distillation was hidden from public view, but today it is widespread and used throughout many industries, from, spirits to the gasoline in your automobile.
http://en.wikipedia.org/wiki/File:Zosimosapparat.jpg
Distillation played an important in the development of early sciences, just as it plays vital role in the manufacturing of thousands of products today. In the early days of alchemy, distillation was hidden from public view, but today it is widespread and used throughout many industries, from, spirits to the gasoline in your automobile.
The earliest forms of distillation involved containers known as Retorts, which are small glass vessels with long downward-pointing spouts, which taper towards the end. The process of using Retorts to distill is a faily simple one. The design allows for the bowl of the retort to be placed upon a heat source, and the heat causes the elements to separate into vapor, which then recondenses back into a liquid.
Equipment
Most distilleries you find online are actually homemade systems and not available for purchase by the average person. There are many regulations and laws regarding still construction and use, especially when it comes to "alcohol". There is some flexibility, however, in our regulations regarding homemade systems, at least until they find a way, right!
If you search long enough you find hundreds if not thousands of designs, God bless our desire to distill things, right! Please consider first that stills are dangerous, even explosive if not constructed, maintained, and operated correctly. The main culprit of this hazard is steam and pressure, which build up as liquids turn to vapor and expand. My favorite is the Percolator Distillery. Open-fire Percolators are found in any outdoor department. I prefer the percolator as a means to distill water because the spout allows you to add more water as you distill, without moving or touching the lid, which can lead to contamination.
If you search long enough you find hundreds if not thousands of designs, God bless our desire to distill things, right! Please consider first that stills are dangerous, even explosive if not constructed, maintained, and operated correctly. The main culprit of this hazard is steam and pressure, which build up as liquids turn to vapor and expand. My favorite is the Percolator Distillery. Open-fire Percolators are found in any outdoor department. I prefer the percolator as a means to distill water because the spout allows you to add more water as you distill, without moving or touching the lid, which can lead to contamination.
Percolator Distillery
I have a specific method I use to distill water using a Percolator. This method is fairly easy, less hazardous than conventional designs, and a good way to distill small amounts of water. Eventually I will be adding larger homemade units to this section, but I absolutely want to provide a minimum of what you could use right now, to survive.
What You Will Need!

You will need:
A Percolator (Of Course), whatever size you want it really does not matter. Remove Coffee basket and tube and set aside.
A Large Strip of Foil, this will be used to make a cradle to hold the cup.
A small cup that fits inside the percolator.
Water and Heat Source
A Percolator (Of Course), whatever size you want it really does not matter. Remove Coffee basket and tube and set aside.
A Large Strip of Foil, this will be used to make a cradle to hold the cup.
A small cup that fits inside the percolator.
Water and Heat Source
Step One

Step One: Remove a long piece of foil from the roll and fold over length wise, until it is about two inches wide. Make the cradle by placing the foil inside the percolator, over-lapping on each side. Note: Your foil should be above where your intended water line will be. You absolutely do not want you foil and cup sitting in your untreated water. The next part of this step is to press the foil against the outer rim of the percolator to keep it from slipping into the percolator. Foil catches very well when pressed tightly against another object. If you folded the foil over several times you should also have eliminated any chance of it tearing.
Step Two

Step Two: Sit the cup gently into the cradle to test for support, do not add water until you have tested the cradle. Remove cup and sit the percolator on fuel source, replace cup.
Step Three

Step Three: Add water to through the spout on the percolator to keep it untreated water from contacting your reservoir (cup inside percolator). Next Place the lid back on the percolator in the UPSIDE DOWN position. Water vapor will condense on the lid, trickle down to the nob on the lid and drip into the cup. Caution: Your best bet is to bring the water to a slow boil, and not a rapid boil. You absolutely do not want untreated water bubbling into your cup.
Step Four
Continue checking reservoir (cup) inside the percolator about every five minutes or so. You can continue to add water and remove the distilled as you go. It takes a little patience and tiny fingers to remove the cup. Do not get into a big rush to get the water, if the cup falls back into the soup below than you will have to start all over. You could even use a straw to drink the collected water. Caution: Whatever chemicals your removing from the water will concentrate in the bottom of the percolator, which could be nocuous gases, be careful.
Continue checking reservoir (cup) inside the percolator about every five minutes or so. You can continue to add water and remove the distilled as you go. It takes a little patience and tiny fingers to remove the cup. Do not get into a big rush to get the water, if the cup falls back into the soup below than you will have to start all over. You could even use a straw to drink the collected water. Caution: Whatever chemicals your removing from the water will concentrate in the bottom of the percolator, which could be nocuous gases, be careful.


